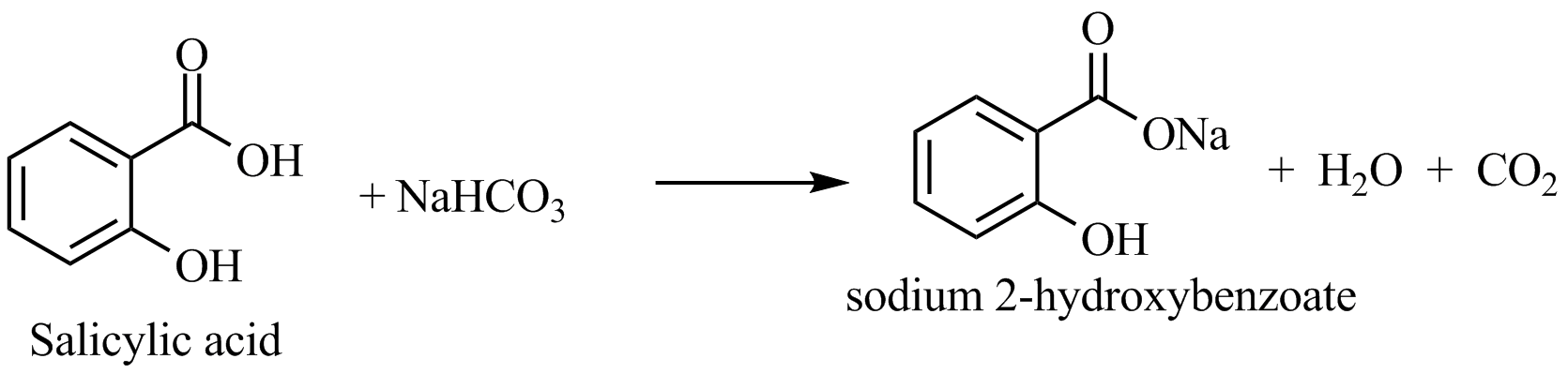
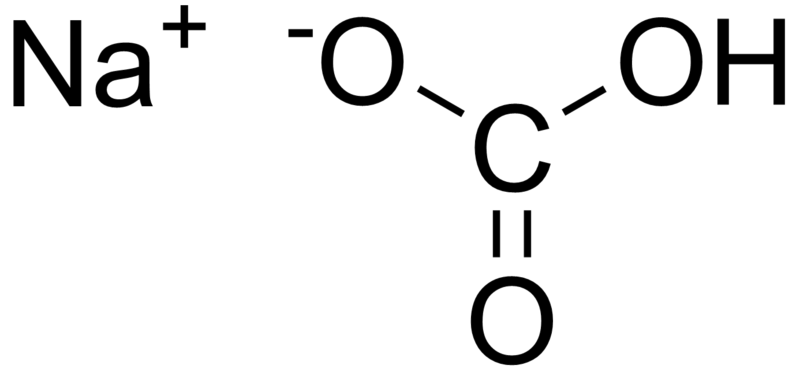
Download top and best high-quality free Sodium Bicarbonate PNG Transparent Images backgrounds available in various sizes. To view the full PNG size resolution click on any of the below image thumbnail.
License Info: Creative Commons 4.0 BY-NC
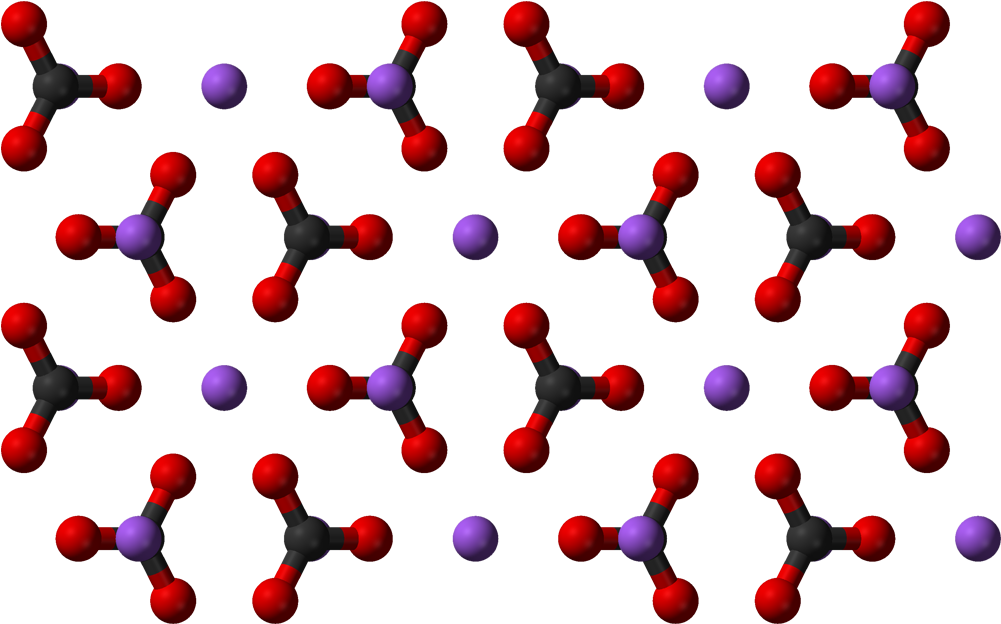
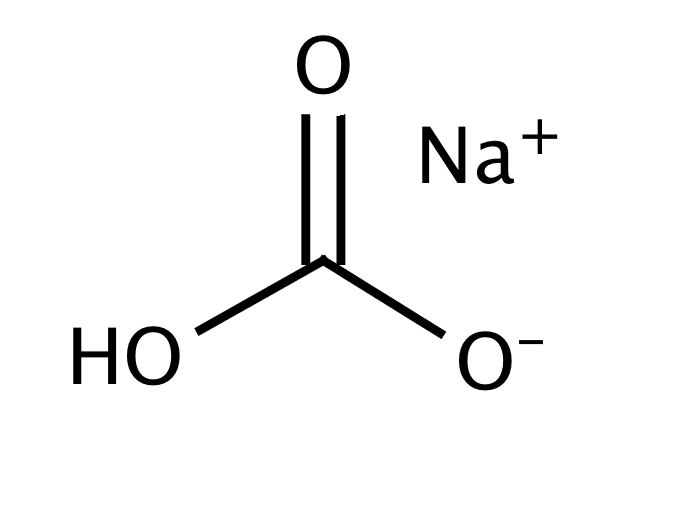
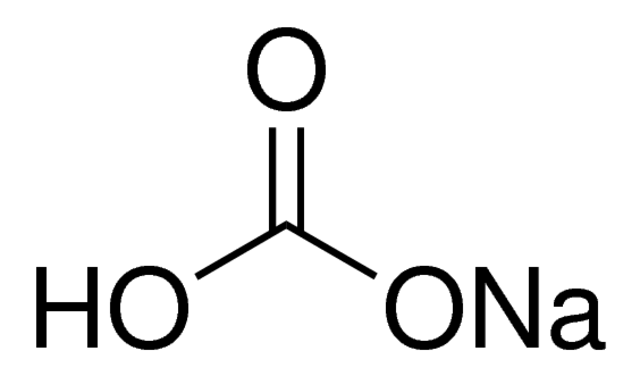
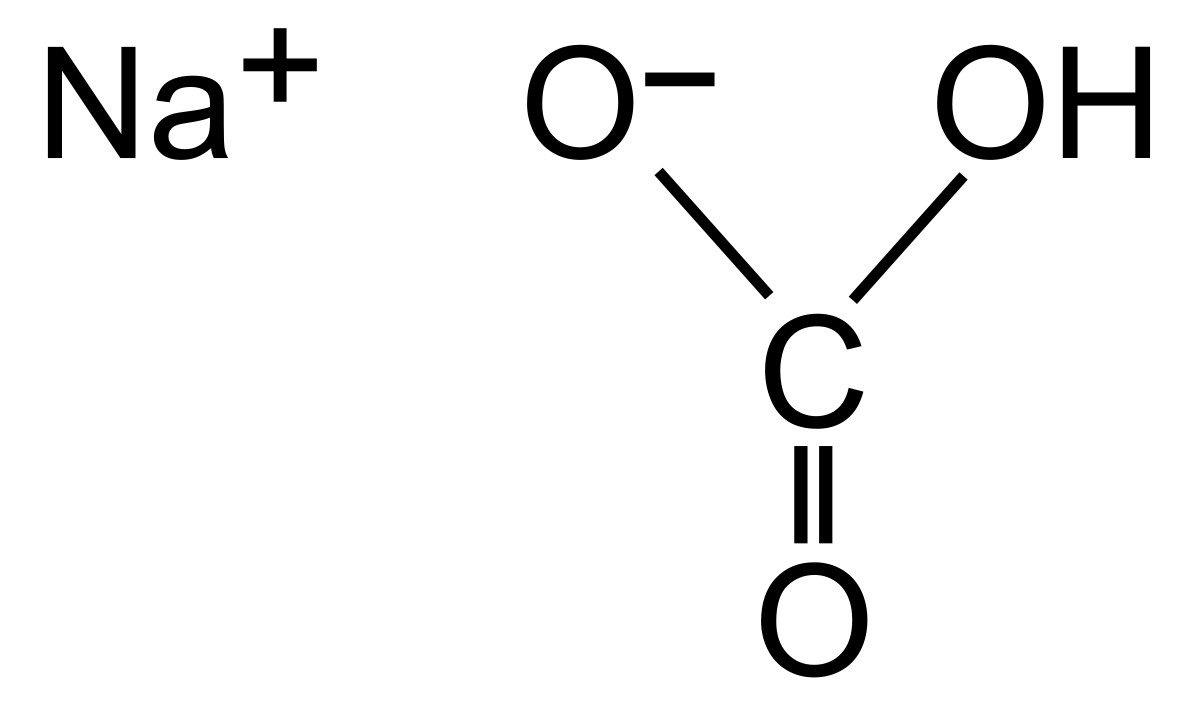
Sodium bicarbonate (IUPAC name: sodium hydrogen carbonate), commonly known as bakingsoda (especially in North America and New Zealand) or baking soda, is a chemical compound of the formula NaHCO3. It is a salt composed of a sodium cation (Na+) and bicarbonate anion (HCO3-). Sodium bicarbonate is a white solid that is crystalline but often in the form of a fine powder. It has a slightly salty, alkaline taste similar to that of washing soda (sodium carbonate). The natural mineral form – is nahcolite. It is part of the mineral natron and is dissolved in many mineral springs.
Because salt has long been known and widely used, it has many associated names such as baking soda, bread soda, cooking soda, and bicarbonate of soda. The term “baking soda” is more common in the United States, whereas bicarbonate of soda is more common in Australia and Britain. In colloquial speech, the names sodium bicarbonate and sodium bicarbonate are often abbreviated; forms such as sodium bicarbonate, baking soda, bicarbonate, and bicarb are common.
The word saleratus, from the Latin sal æratus (meaning “aerated salt”), was widely used in the 19th century for sodium bicarbonate and potassium bicarbonate. It is known as one of the food supplements with the E number E500.
The bi prefix in bicarbonate comes from an outdated naming system and is based on the observation that sodium bicarbonate (NaHCO3) contains twice as much carbonate (CO3) per sodium as sodium carbonate (Na2CO3). The modern chemical formulas of these compounds express their exact chemical composition (which was not known when the names sodium carbonate and sodium bicarbonate were introduced), such as sodium hydrogen carbonate (NaHCO3) and sodium carbonate (Na2CO3). These names are unambiguous since sodium always has an oxidation state of +1 and carbonizes an oxidation state of -2.
Sodium bicarbonate was sometimes used in the preparation of green vegetables because it gives them a bright green color – which is described as artificial – due to its reaction with chlorophyll to form chlorophyllin. However, it does affect flavor, texture, and nutritional value and is no longer found.
Baking soda is used to soften legumes (peas, beans) before and during cooking, like the traditional British mushy peas recipe for soaking peas. The main effect of sodium bicarbonate is to change the pH of the acidifying solution and the cooking water; this softens the hard outer shell, shortens cooking times, and can change the nutrient percentage of the dish, its flavor, and texture. Soda can react with acids in food, including vitamin C (L-ascorbic acid).
Download Sodium Bicarbonate PNG images transparent gallery.
- Sodium Bicarbonate Chemical Compound PNG Free Download
Resolution: 1100 × 982
Size: 138 KB
Image Format: .png
Download
- Sodium Bicarbonate Formula PNG
Resolution: 2069 × 1349
Size: 338 KB
Image Format: .png
Download
- Sodium Bicarbonate Formula PNG Image
Resolution: 2256 × 1592
Size: 380 KB
Image Format: .png
Download
- Sodium Bicarbonate
Resolution: 520 × 641
Size: 321 KB
Image Format: .png
Download
- Sodium Bicarbonate PNG Free Download
Resolution: 591 × 406
Size: 364 KB
Image Format: .png
Download
- Sodium Bicarbonate Chemical Compound
Resolution: 1094 × 767
Size: 194 KB
Image Format: .png
Download
- Sodium Bicarbonate PNG Picture
Resolution: 505 × 448
Size: 300 KB
Image Format: .png
Download
- Sodium Bicarbonate PNG Free Image
Resolution: 600 × 600
Size: 492 KB
Image Format: .png
Download
- Sodium Bicarbonate PNG File
Resolution: 1080 × 1080
Size: 708 KB
Image Format: .png
Download
- Sodium Bicarbonate PNG HD Image
Resolution: 1024 × 1024
Size: 1186 KB
Image Format: .png
Download
- Sodium Bicarbonate PNG Pic
Resolution: 1000 × 1000
Size: 243 KB
Image Format: .png
Download
- Sodium Bicarbonate Chemical Compound PNG
Resolution: 1001 × 624
Size: 226 KB
Image Format: .png
Download
- Sodium Bicarbonate PNG Download Image
Resolution: 800 × 714
Size: 204 KB
Image Format: .png
Download
- Sodium Bicarbonate PNG High Quality Image
Resolution: 1114 × 780
Size: 289 KB
Image Format: .png
Download
- Sodium Bicarbonate PNG
Resolution: 1200 × 819
Size: 1128 KB
Image Format: .png
Download
- Sodium Bicarbonate Formula Transparent
Resolution: 696 × 520
Size: 9 KB
Image Format: .png
Download
- Sodium Bicarbonate PNG Image
Resolution: 865 × 450
Size: 396 KB
Image Format: .png
Download
- Sodium Bicarbonate Formula PNG Clipart
Resolution: 1671 × 399
Size: 16 KB
Image Format: .png
Download
- Sodium Bicarbonate Chemical Compound PNG Image
Resolution: 1100 × 723
Size: 250 KB
Image Format: .png
Download
- Sodium Bicarbonate Formula PNG Free Download
Resolution: 800 × 369
Size: 39 KB
Image Format: .png
Download
- Sodium Bicarbonate Formula PNG Picture
Resolution: 1200 × 659
Size: 12 KB
Image Format: .png
Download
- Sodium Bicarbonate Chemical Compound Transparent
Resolution: 944 × 768
Size: 111 KB
Image Format: .png
Download
- Sodium Bicarbonate Transparent
Resolution: 500 × 500
Size: 430 KB
Image Format: .png
Download
- Sodium Bicarbonate PNG Clipart
Resolution: 700 × 489
Size: 144 KB
Image Format: .png
Download
- Sodium Bicarbonate Formula
Resolution: 640 × 376
Size: 13 KB
Image Format: .png
Download
- Sodium Bicarbonate Chemical Compound PNG Clipart
Resolution: 1100 × 1099
Size: 218 KB
Image Format: .png
Download
- Sodium Bicarbonate Formula PNG Free Image
Resolution: 1200 × 702
Size: 16 KB
Image Format: .png
Download